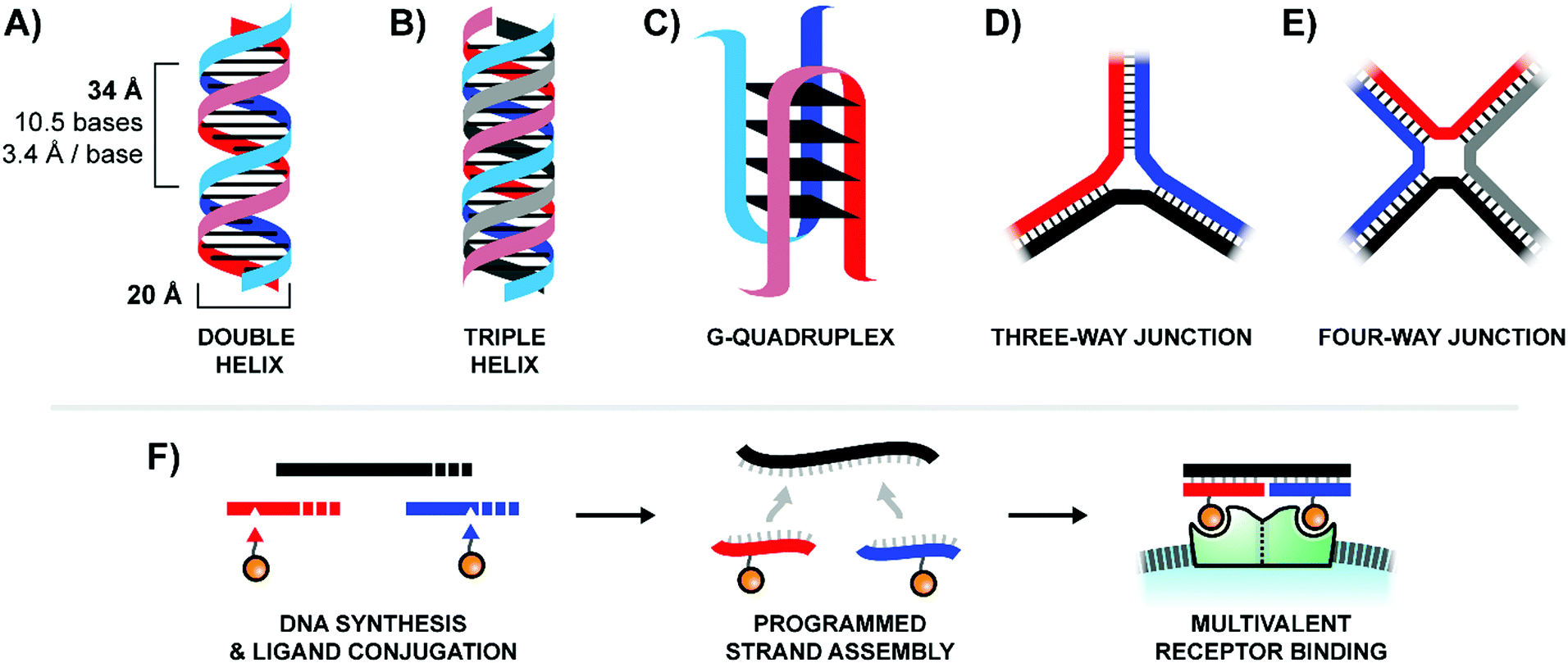
The collabody erb_scFv-Col bound to the extracellular domain of epidermal growth factor receptor with a binding strength approximately 20- and 1000-fold stronger than the bivalent and monovalent counterparts, respectively. The collagenous motif in collabody is prolyl-hydroxylated, with remarkable thermal and serum stabilities. The collabody consisting of scFv fused to the N terminus of collagen scaffold is present as a homotrimer, whereas it exhibited a mixture of trimer and interchain disulfide-bonded hexamer when cysteine residues were introduced and flanked the scaffold. 
Different forms of collabody, consisting of the human single-chain variable fragment (scFv) fused to either the N or C terminus of the collagen-like peptide scaffold (Gly-Pro-Pro)(10), were stably expressed as soluble secretory proteins in mammalian cells. These binders, termed "collabodies," use a triplex-forming collagen-like peptide to drive the trimerization of a heterologous target-binding domain. We use a combination of molecular biology, biochemistry, optogenetics and high-resolution microscopy to understand the mechanisms, the internal organization and the functions that arise specifically from the assembly of membrane-less compartments in the DDR.A class of multivalent protein binders was designed to overcome the limitations of low-affinity therapeutic antibodies. For example, we reported recently that the multivalent protein scaffold TOPBP1 drives the assembly of nuclear foci to activate ATR/Chk1 signaling. These cooperative and reversible interactions promote the formation of micron-sized protein networks that fulfill the functions of subcellular compartments. Foci formation is triggered by posttranslational modifications that increase attractive interactions. Our working model is that the recruitment of a key multivalent protein scaffolds at DNA lesions nucleates the assembly of DDR foci. We consider DDR nuclear foci as membrane-less compartments that control the spaciotemporal organization of DNA damage responses. Proteins required for the detection, the signaling and the repair of DNA lesions typically accumulate within distinct nuclear sites visualized as foci by immunofluorescence staining. In recent years, our main focus has been on the mechanisms and functions of compartmentalization in the DNA damage response (DDR). We have analyzed systematically the protein composition of replication sites in basal conditions and in response to a variety of chemotherapeutic agents to identify proteins that ensure the progression of replication forks. These mechanisms are determinants for tumor growth and resistance to chemotherapies. Our objective is to understand the biochemical mechanisms that underpin cellular responses to DNA lesions. Therefore, cancer cells rely on DNA damage responses to sustain growth in the presence of a high load of endogenous lesions and during chemotherapeutic treatments. Furthermore, metabolic rewiring associated with carcinogenesis induces DNA lesions. Inherited defects in the mechanisms of genome maintenance is the underlying cause of cancer prone disorders such as Fanconi anemia, a disease characterized by defects in the repair of inter-strand DNA crosslinks and in the rescue of stalled replication forks. Therefore, all living organisms have evolved diverse mechanisms to repair alterations in the primary structure of DNA. DNA repair mechanisms are crucial for organismal health and survival.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
